Improving Data Integrity: Understanding the ALCOA and ALCOA Plus Principles
Discover the essentials of data integrity with our guide on the ALCOA and ALCOA Plus principles. Learn how these standards ensure data is Attributable, Legible, Contemporaneous, Original, and Accurate, with extensions like Complete, Enduring, Available, and Consistent, crucial for compliance in regulated industries. Enhance your understanding and implementation of best practices for maintaining high-quality data integrity.
-
Introduction
Data integrity is essential in the pharmaceutical industry, directly impacting the safety, efficacy, and reliability of pharmaceutical products. The ALCOA and ALCOA Plus Principles are essential for ensuring data accuracy and reliability, helping to reduce risk and comply with regulatory requirements.
-
Understanding the ALCOA and ALCOA Plus Principles
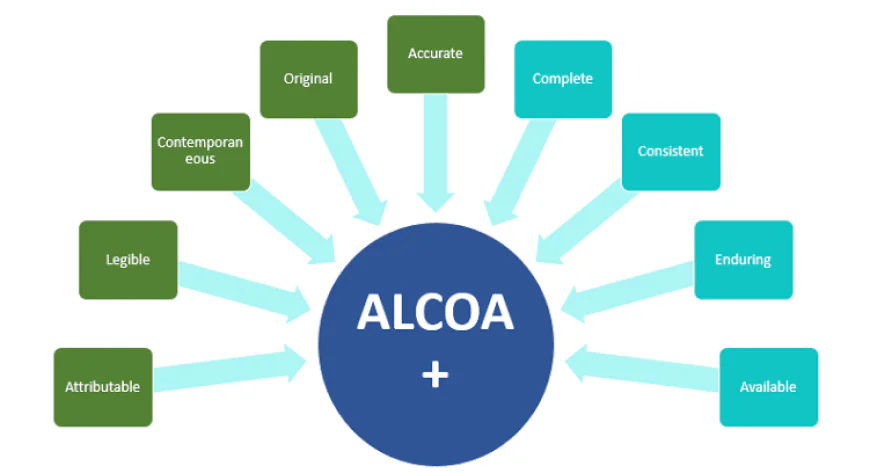
The ALCOA principles (attributable, legible, synchronized, authentic, and accurate) were designed as a framework for improving data integrity in the pharmaceutical industry. These principles were later expanded to become ALCOA Plus, adding the new concepts: complete, consistent, permanent, and available. These principles are recognized by regulatory bodies such as the FDA and WHO, underscoring their importance in ensuring data reliability.
-
ALCOA Principles
1. Attributable: Data must be traceable to the person who collected it, with a specific time and location.
2. Legitimacy:Data must be clear and legible, whether handwritten or electronic.
3. Synchronized: Data must be recorded in real time to ensure accuracy.
4. Original: Maintaining original records is essential to maintaining data integrity.
5. Accurate: Data must be error-free, and any modifications must be supported by documentation. -
Additional Principles of ALCOA Plus
1. Complete: Data must be complete, with no missing parts.
2. Consistent: Data must be organized in a chronological and logical order.
3. Permanent: The media used to store data must be capable of being retained for a long period of time.
4. Available: Data must be readily available and accessible when needed. -
Importance Across Industries
While the ALCOA and ALCOA Plus principles originated in the pharmaceutical sector, they have been widely adopted in other industries such as healthcare and food production. In these industries, these principles ensure document integrity and product safety.
-
Practical Applications
Implementing the ALCOA and ALCOA Plus principles requires strong documentation practices, regular audits, and employee training. Companies can use modern software solutions to facilitate accurate and efficient data collection and storage.
-
Conclusion
In conclusion, the ALCOA and ALCOA Plus principles are vital tools for ensuring data integrity in various industries. By understanding and applying these principles, companies can improve their data management practices, leading to more reliable products, improved decision-making, and compliance with regulatory requirements. Adopting these principles not only reduces risk but also enhances trust and credibility, which are essential elements for long-term success in today's competitive marketplace.







